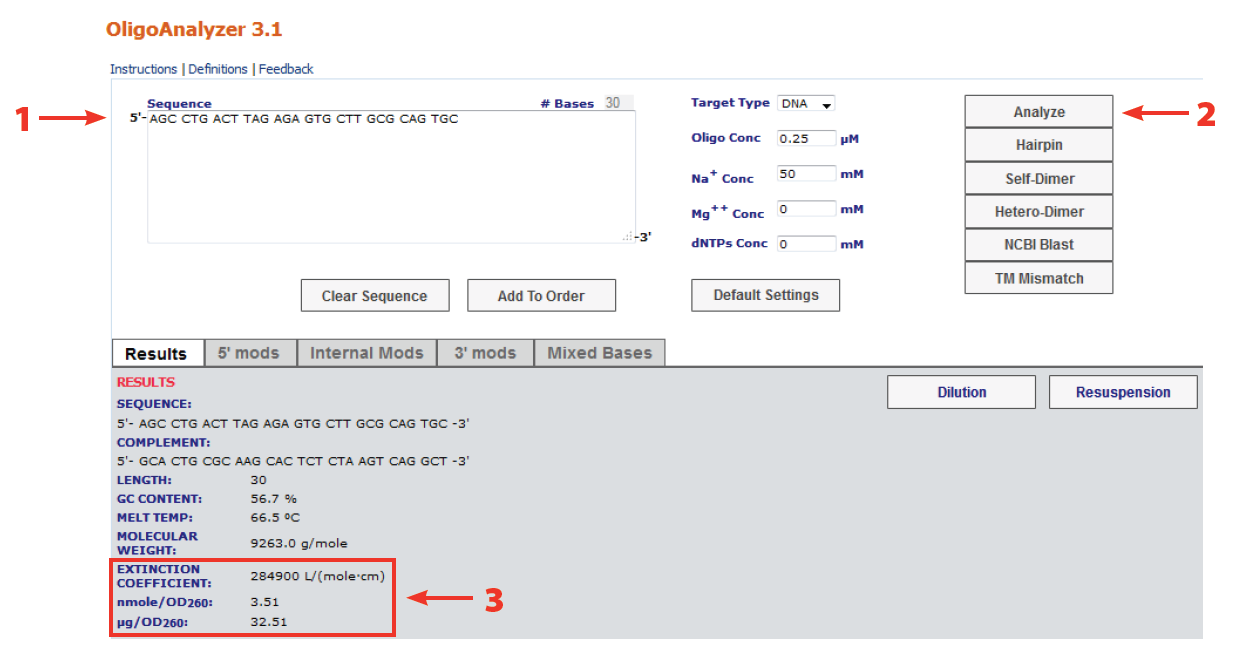
How to measure extinction coefficient of para nitrophenyl acetate (pH 7.6 tris sulphate buffer; temperature 25 degree)? | ResearchGate
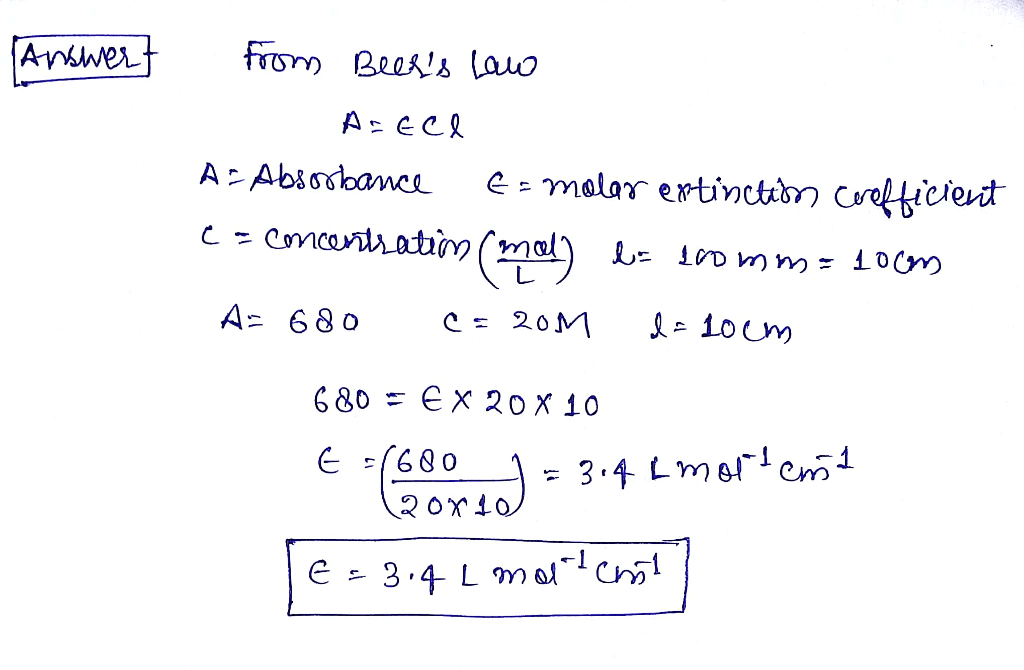
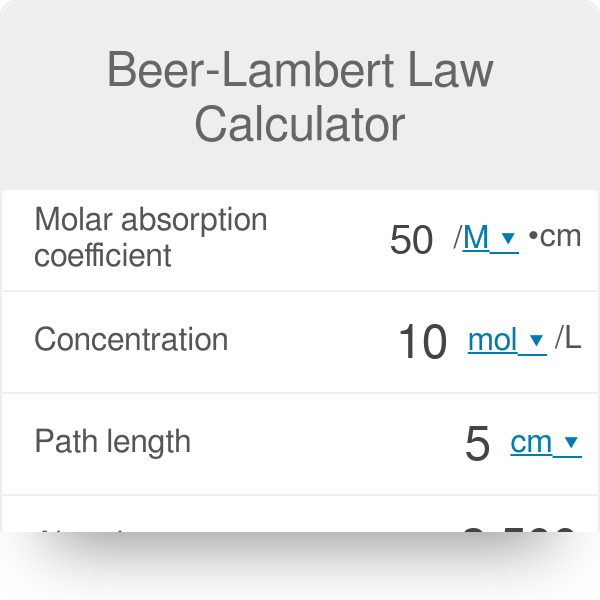
OneClass: What is the molar extinction coefficient of a compound (20 moles/L) that has an absorbance ...

Spectrophotometric determination of ozone in solutions: Molar absorption coefficient in the visible region - ScienceDirect

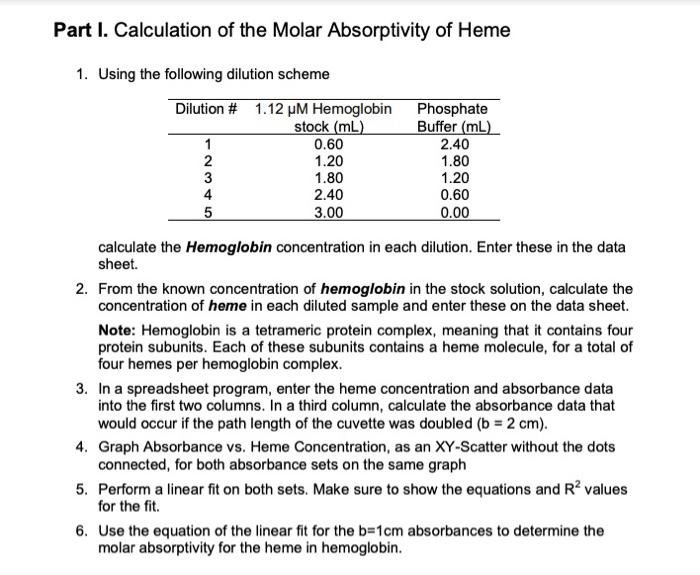
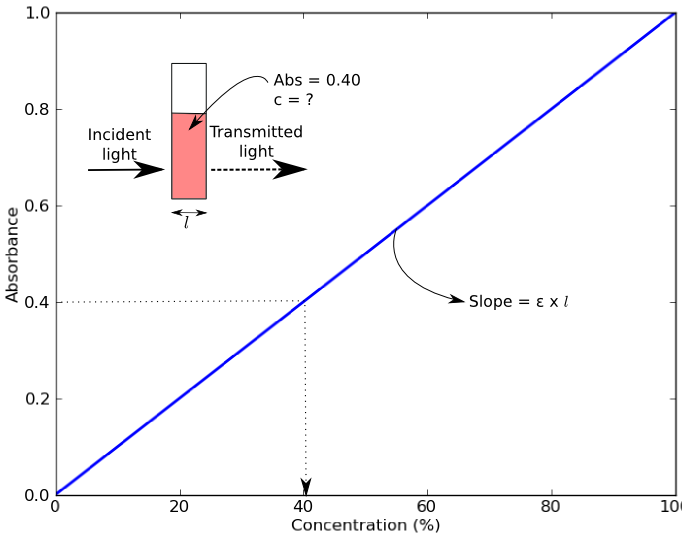
SOLVED: Molar Absorptivity Absorbance is directly proportional to the path length, b, and the concentration, C of the absorbing species. Beer's Law states that A = cl From Spectra A is the