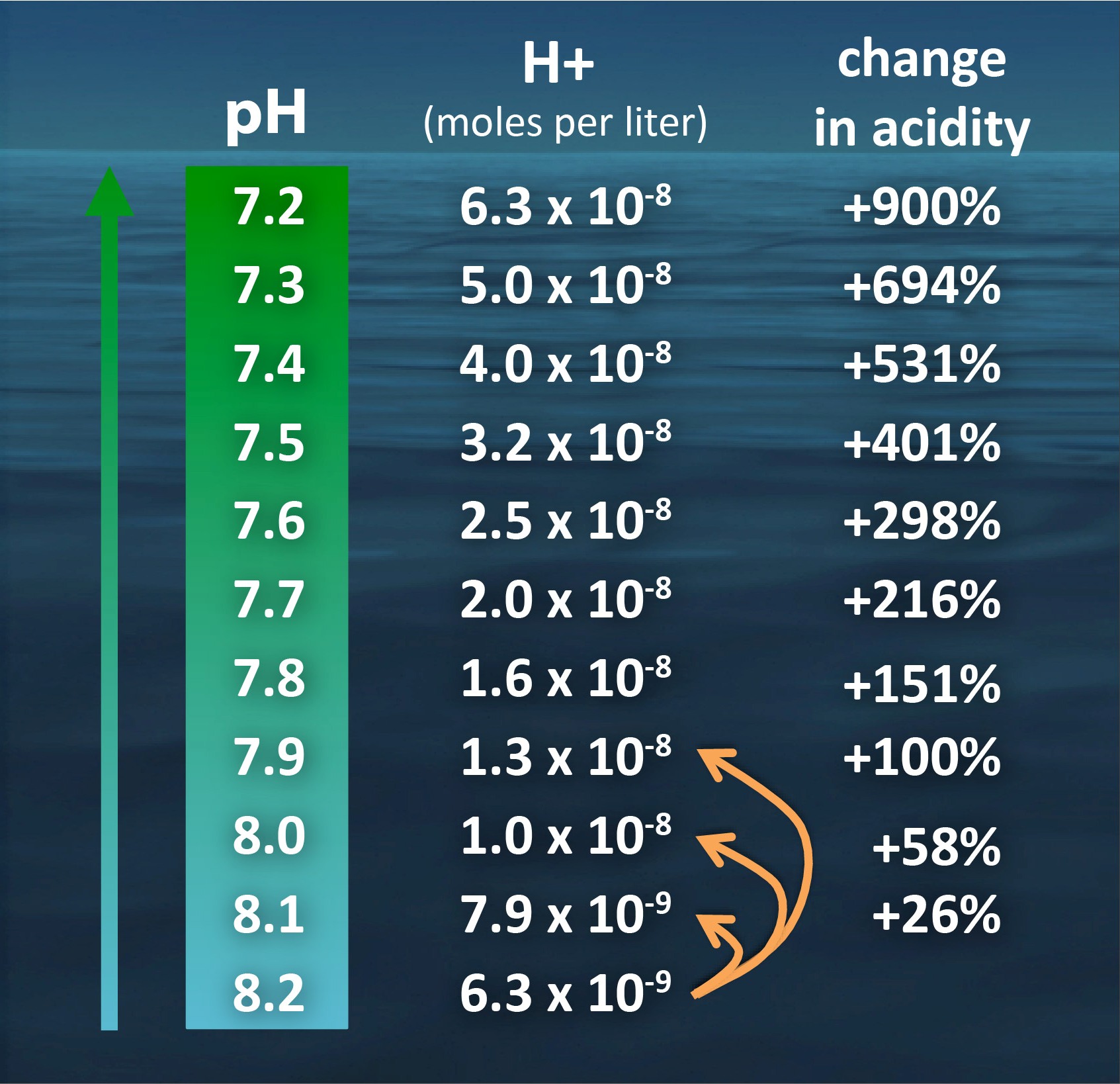
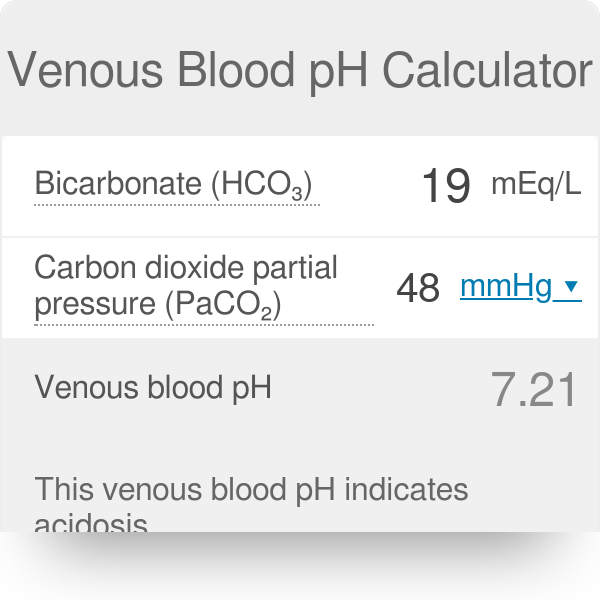
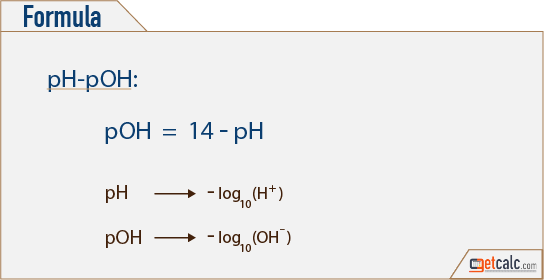
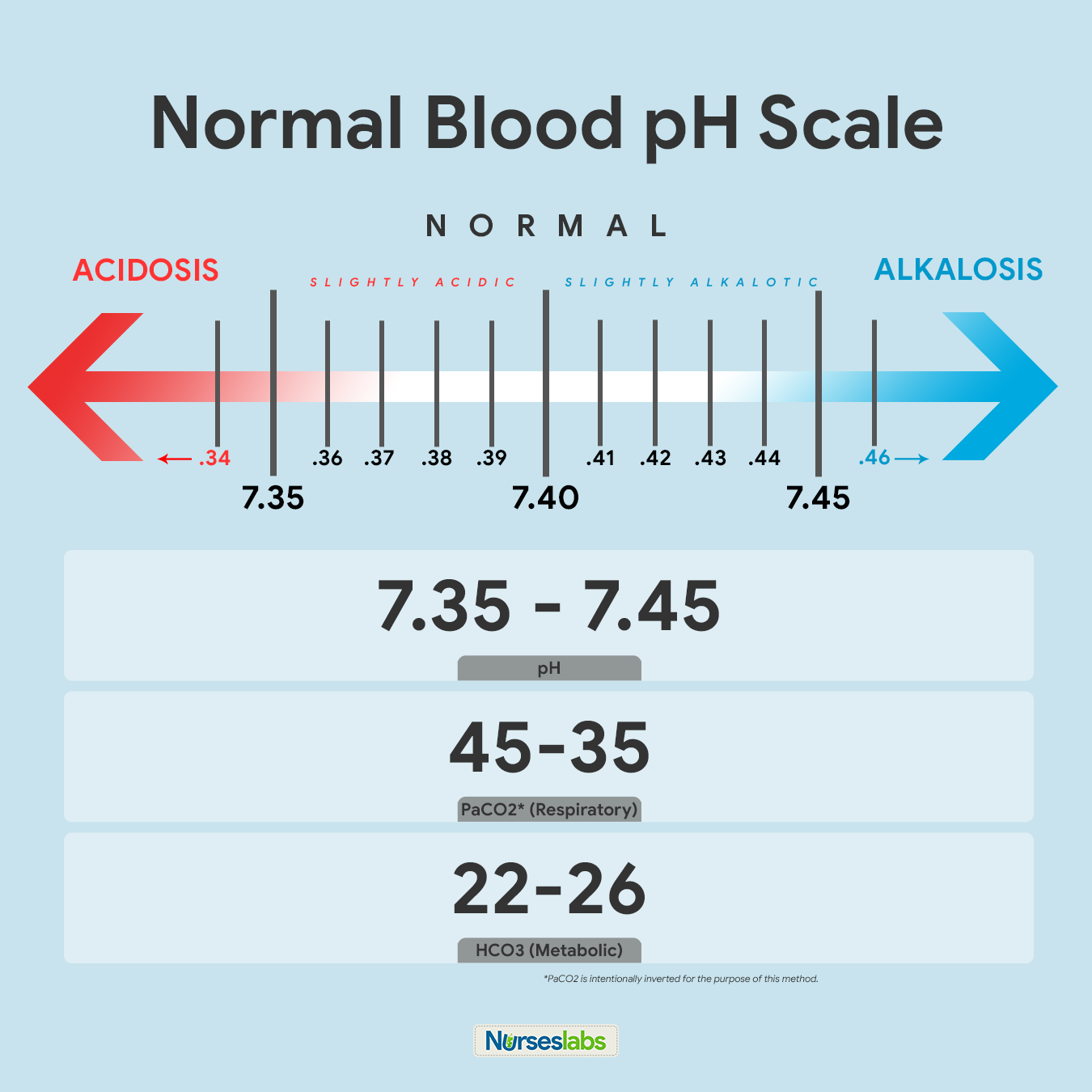
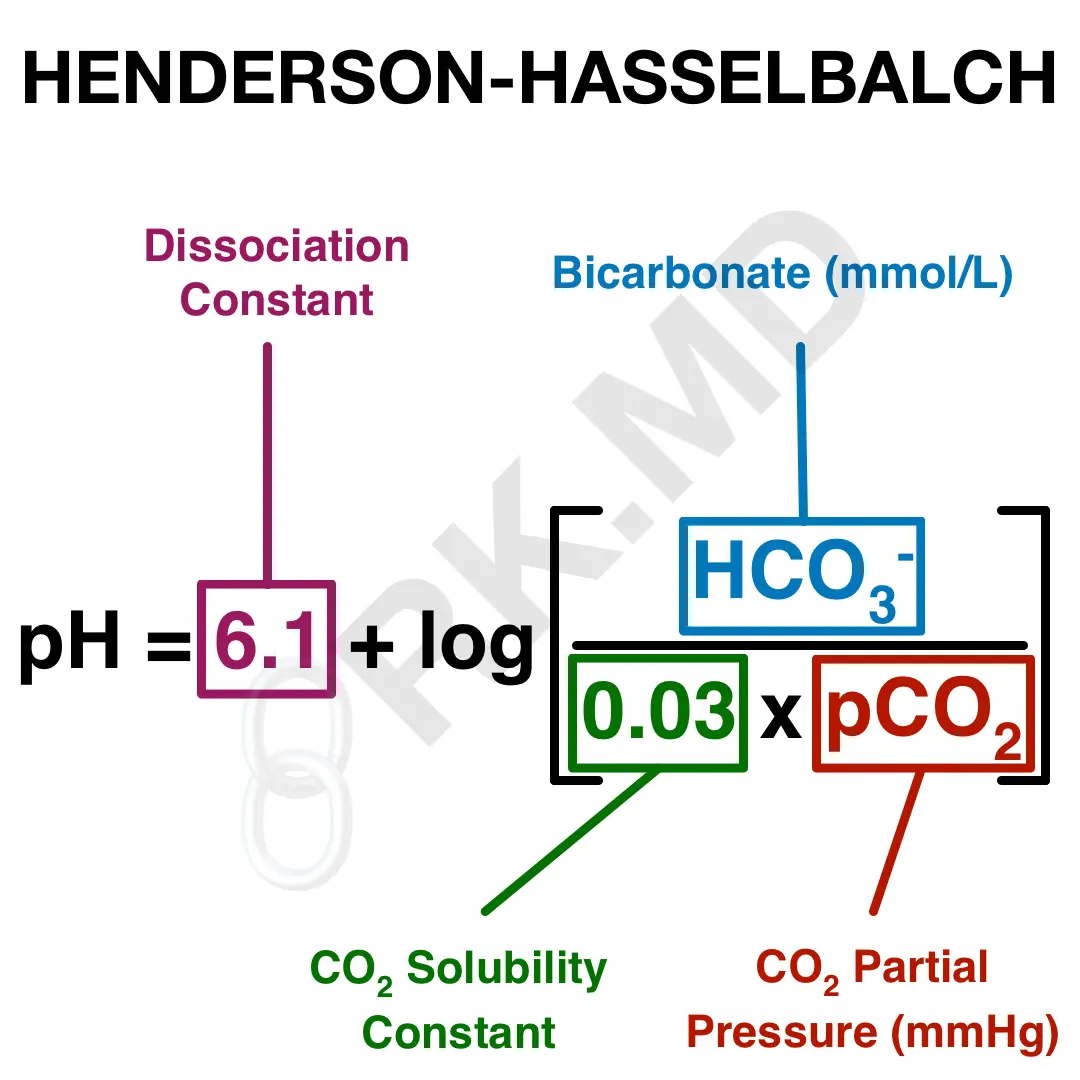
SOLVED: Calculate the pH of a blood plasma sample with a total CO2 concentration of 26.9 mM and bicarbonate concentration of 25.6 mM. Recall from page 67 that the relevant pK2 of

The approximate molar concentration of several chemicals are given. Find the pH of each use the calculator - brainly.com
![The `pH` of blood is 7.4 . What is the ratio of `[(HPO_(4)^(2-))/(H_(2)PO_(4)^(-))]` in the blood. ` - YouTube The `pH` of blood is 7.4 . What is the ratio of `[(HPO_(4)^(2-))/(H_(2)PO_(4)^(-))]` in the blood. ` - YouTube](https://i.ytimg.com/vi/irWUJj_qIt8/maxresdefault.jpg)